
Derma Veil is a POLYLACTIC ACID, non-toxic, bioabsorbable and biodegradable tissue reconstructive material to treat moderate to severe wrinkles
WHAT IS DERMA VEIL?
Derma Veil is a lyophilized low viscosity, non-toxic, bioabsorbable and biodegradable tissue reconstructive material with the following characteristics:
- Disposable single use medical device incorporated as a hydrophilic matrix. Its active principle is composed of glycolic acid (GA) bound to the polymeric column of polylactic acid [PURASORB® Poly (L-lactide)] both of which are homogeneously dispersed throughout and their rate of release is controlled by diffusion, prolonging a sustained repairing action through the slow dissolution of the compound.
- Immunologically inactive, biocompatible and absorbable and is degraded by hydrolysis. The degradation begins first by water diffusion in the material, initially at the more amorphous zones, followed by hydrolysis, material fragmentation and finally, a more extensive hydrolysis along with phagocytosis, diffusion and metabolization. Elimination occurs primarily through the respiratory tract.
Each box of Derma Veil contains 2 vials and an Instruction for Use leaflet. The vials are made of transparent borosilicate glass, sealed with a siliconized halobutyl stopper secured by an aluminum ring with a "flip off" plastic cap.
The vial has 10 mL capacity and contains 236.14 mg of a lyophilized, sterile, pyrogen free, crystalline white powder of asymmetric micro-particles made of:
- - POLYLACTIC ACID a biocompatible, biodegradable synthetic polymer for medical applications, [PURASORB® Poly (L-lactide)]
- - GLYCOLIC ACID a biocompatible, biodegradable, high purity hydroxyacetic acid monomer for medical applications,
- - CARBOXY METHYL CELLULOSE SODIUM (USP),
- - MANNITOL (USP),
- - POLYSORBATE 80 (NF),
INTENDED USE:
Derma Veil is applied by subdermal use to treat moderate to severe facial fat loss (lipoatrophy) caused by degenerative changes that occur due to illness or advanced age.
Its application triggers a foreign body reaction promoting a glucosaline biosynthesis that increases cellular interactions favoring the production of new collagen. The process generates histological changes that provide greater fullness to the cutaneous tegument.
These functions provide reconstruction to collapsed areas while diminishing skin depressions such as wrinkles, creases and minor scars.
- - The treatment requires a minimum of two sessions separated by 3 weeks (20 days).
- - Derma Veil is activated prior to use by adding preferably 8mL of Physiological Saline Solution (0.91% w/v of NaCl, about 300 mOsm) or 8mL of Sterile Water for use (pyrogen and preservative free)
Once activated, the formula becomes a suspension of relative viscosity.
- - Derma Veil should be delivered using a sterilized 26G½ (0.45mm x 13mm)
STORAGE:
Keep Derma Veil in dry storage, away from humidity and light, at a temperature between 15º C (59º F) and 30º C (86º F). Refrigeration is not necessary.
CONTRAINDICATIONS:
- - Derma Veil should not to be used in patients with hypersensitivity or allergy to any of the components in the formula.
- - Derma Veil should not to be used in patients with a history or presence of severe allergies.
- - Derma Veil should not to be used if a dermatological condition or an active inflammatory process or infection is present
- - The safety of Derma Veil for use in women who are pregnant, suspected of being pregnant or who are lactating has not been established
- - Do not use Derma Veil in woman's breast region due to potential imaging interference during diagnostic procedures
- - Do not use Derma Veil in patients who have a history or susceptibility to hypertrophic scarring or keloid formation.
- - Do not use Derma Veil in patients with bleeding disorders or patients who are using medications that can prolong bleeding, such as Heparin, Plavix or aspirin
- - Do not use Derma Veil if other fillers have been used within six (6) months prior to a programmed treatment and never use Derma Veil if silicone or methacrilate fillers are present
CONTRAINDICATIONS:
- - Derma Veil is only for the use of licensed physicians familiar with disposable fillers
- - Derma Veil can only be used for dermal application. Never apply the product in the epidermis (superficially) or in subcutaneous tissue.
- - Caution when using Derma Veil in areas where the tissue is thin such as; temples, glabella, lateral orbital rim (crow's feet). These applications require skill and well developed practice.
- - The safety of Derma Veil for use in sub - orbital region has not been studied or evaluated and should be avoided.
- - The safety of Derma Veil for use in the area of the lips (vermillion) has not been studied or evaluated and should be avoided.
- - Do not overfill a contour deficiency because the depression should slowly diminish as the benefits of the product become evident during the following weeks after the sessions.
- - The following reactions to the use of local anesthesia and/or Derma Veil during/after a treatment session have been observed consisting mainly of short-term (i.e., a few hours to 5 days) duration: ecchymosis, erythema, edema, hematoma, and bleeding.
- - Patients have expressed various levels of discomfort/pain caused by the use of Derma Veil, as well as due to the devices presence refer to the Clinical Studies
- - Do not use Derma Veil in the presence of a dermatological condition, infection or an active inflammatory process such as rashes, hives, and other skin eruptions until the condition has been resolved
- - Do not mix Derma Veil with other products. No studies or analysis have been made to evaluate the interactions of Derma Veil with other substances, drugs, anesthetics, fillers, or prosthesis
- - Do not use Derma Veil in the presence of other dermal fillers delivered within six months prior to a treatment program with Derma Veil.
- -
- - Do not use Derma Veil if a weight control treatment is in progress or projected within four (4) months after a programmed last session of treatment (weight loss may cause volume increase to appear disproportional).
- - The safety and effectiveness of Derma Veil has not been established beyond two years.
- - Each vial of Derma Veil is for single use only.
- - Do not re sterilize Derma Veil
- - Do not freeze Derma Veil.
- - Once the security seal of Derma Veil has been broken the sterility of the device cannot be guaranteed.
- - When receiving Derma Veil do not use the product if the security seal is damaged or has signs of having been tampered with. Report immediately the anomaly to the authorized distributor or to the manufacturer. The product will be replaced as soon as possible.
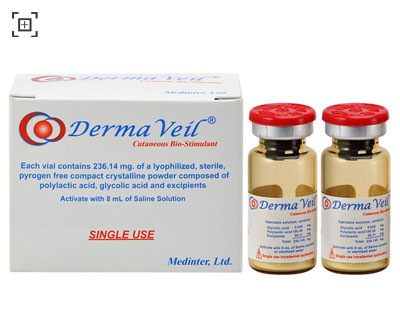
 Derma Veil is a POLYLACTIC ACID, non-toxic, bioabsorbable and biodegradable tissue reconstructive material to treat moderate to severe wrinkles
Derma Veil is a POLYLACTIC ACID, non-toxic, bioabsorbable and biodegradable tissue reconstructive material to treat moderate to severe wrinkles